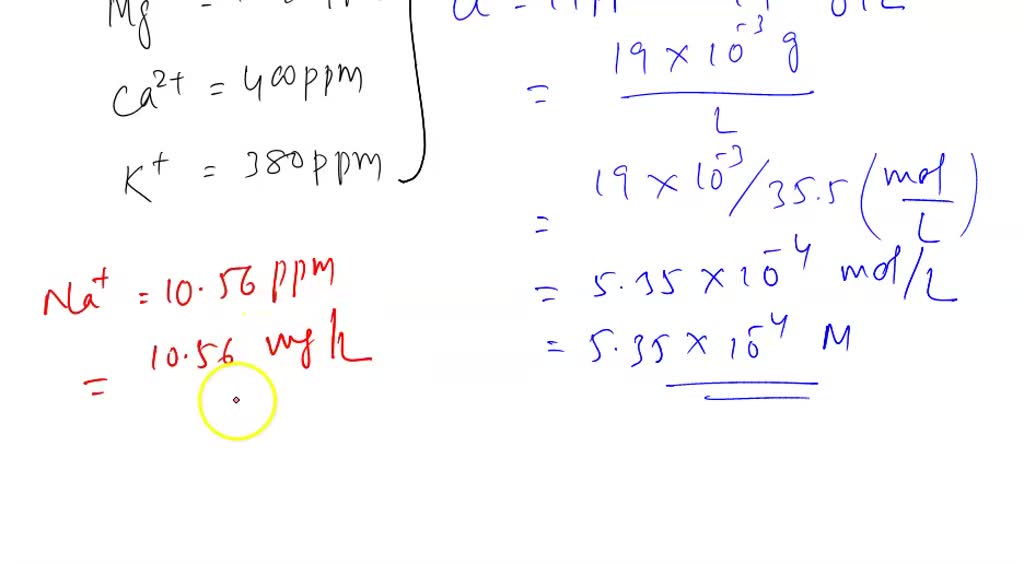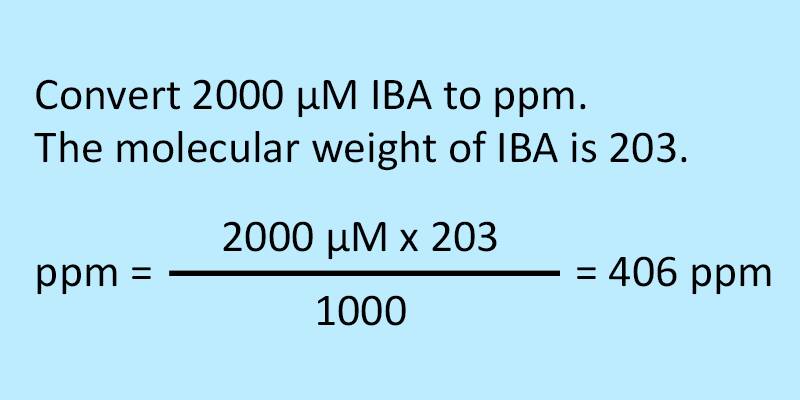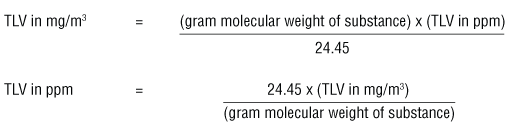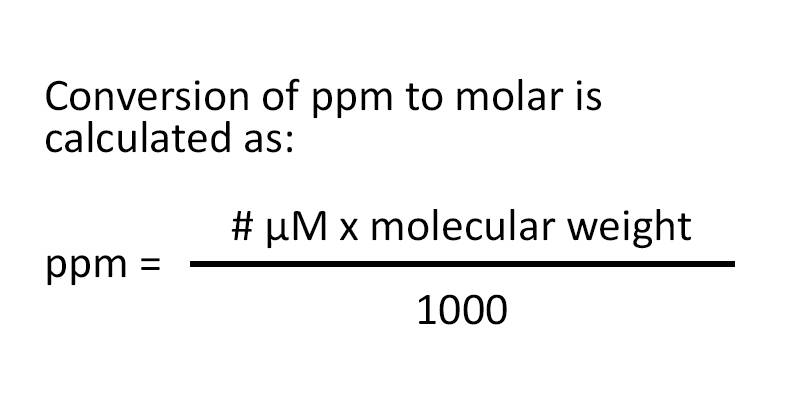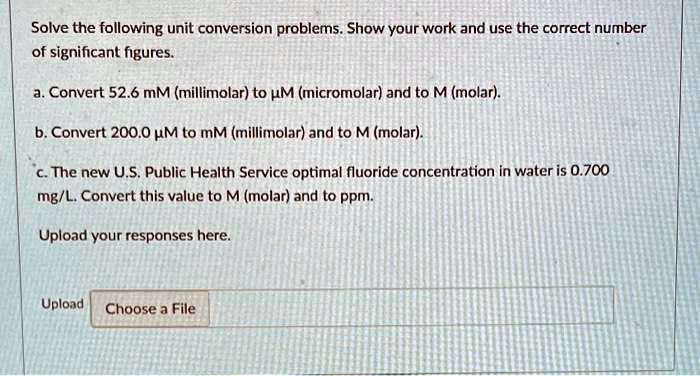
SOLVED: Please help. Solve the following unit conversion problems. Show your work and use the correct number of significant figures. a. Convert 52.6 mM (millimolar) to µM (micromolar) and to M (molar).

SOLVED: 8. Convert 50 ppm to g/L 9. Convert 5 ppm to mL in 100 mL 10. 250 mL of 5 mM CuSO4 MW: 159.6 g/mol 11. 500 mL of 0.2 M

Conversion of CO% at 175 °C over 5.5 mm-sized Cu/ZnO/Al 2 O 3 catalyst... | Download Scientific Diagram

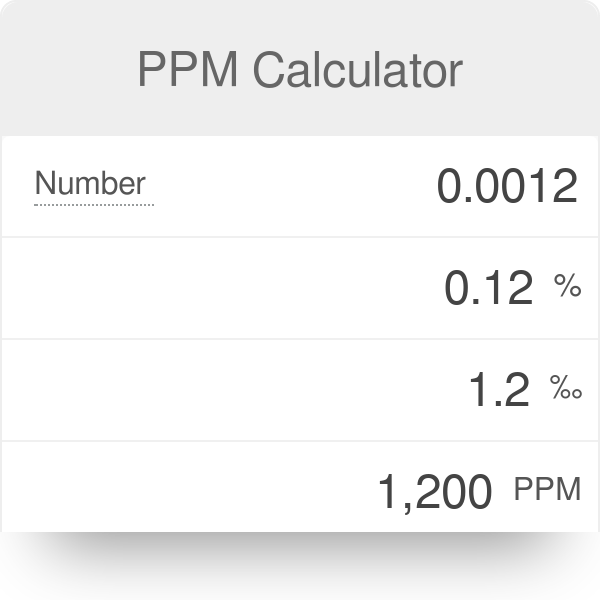
SOLVED: You are given a 300.65 ppm chloride solution. What is the chloride concentration in mmol Cl-/L or mM Cl-? Report your answer to 3 decimal places with no units.